В мире онлайн-ставок приложение 1xBet выделяется как одно из ведущих приложений, предлагающих широкий выбор вариантов ставок на спорт и казино. Это приложение, доступное как для пользователей Android, так и для iOS, обеспечивает удобный доступ к широкому спектру спортивных событий, игр казино, а также к прямым трансляциям и ставкам в реальном времени. Сочетая в себе передовые технологии и пользовательский интерфейс, ориентированный на комфорт пользователя, приложение 1xBet становится не просто платформой для ставок, но и центром азартных развлечений, доступным прямо в вашем кармане.
Возможности приложения 1xBet
| Особенность | Описание |
| Букмекерская контора | Охватывает широкий спектр местных и международных видов спорта с возможностью ставок в реальном времени. |
| Казино игры | Включает игровые автоматы, настольные игры, живое казино и ставки на виртуальный спорт. |
| Пользовательский опыт | Поддержка Русского языка с настраиваемым пользовательским интерфейсом. |
| Финансовые операции | Поддерживает местные способы оплаты |
| Дополнительные возможности | Прямые трансляции событий, push-уведомления и многоязычная поддержка клиентов. |
Приложение 1xBet Казино
- Слоты: обширная коллекция игровых автоматов от различных игровых провайдеров.
- Настольные игры: классические игры казино, такие как блэкджек, рулетка, баккара и покер.
- Живое казино: игры в реальном времени с живыми дилерами для полного погружения в казино.
Мобильный сайт и приложения
| Аспект | Мобильный веб-сайт | Приложение 1xBet |
| Доступность | Нет загрузки, доступен через браузер | Требуется загрузка и установка |
| Пользовательский опыт | Адаптивный дизайн, зависит от браузера | Оптимизированный, плавный и с большим количеством функций |
| Использование данных | Более высокое использование данных на загрузку страницы | Более эффективен после первоначальной загрузки |
| Обновления | Автоматически обновляется | Необходимы обновления вручную |
| Общий | Удобно, не нужно скачивать приложения. | Более быстрый и интегрированный опыт |
Выбор между мобильным сайтом 1xBet и приложением зависит от индивидуальных предпочтений относительно удобства, использования данных и возможностей устройства.

Скачать 1xBet Apk для Android
Для пользователей Android, заинтересованных в ставках с помощью 1xBet, загрузка приложения — это быстрый и простой процесс, который открывает на вашем устройстве мир спортивных ставок и казино. Важно отметить, что для загрузки и установки APK-приложения 1xBet вам необходимо посетить наш веб-сайт 1xBet, поскольку приложение не всегда доступно в магазине Google Play. Вот пошаговая инструкция по загрузке и установке:
- Перейдите на этот веб-сайт: используйте браузер на своем устройстве Android, чтобы открыть веб-сайт 1xBet.
- 1xBet Загрузить APK-файл: Найдите и щелкните ссылку, чтобы загрузить APK-файл приложения 1xBet.
- Разрешить установку: в настройках вашего устройства разрешите установку приложений из неизвестных источников.
- Установите приложение 1xBet: после загрузки APK-файла нажмите на него, чтобы начать установку, и следуйте инструкциям на экране.
Системные требования для Андроид
Для оптимальной работы приложения 1xBet на Android-устройствах должны соблюдаться следующие системные требования:
- Версия Android: 5.0+
- Оперативная память: 2 ГБ
- Процессор: 1,4 ГГц
- Внутреннее пространство: 90 Мб
Скачать приложение 1xBet для iOS
Загрузка приложения 1xBet на устройства iOS — это простой процесс, позволяющий пользователям быстро начать пользоваться широким спектром доступных вариантов ставок. Вот как это сделать:
- Откройте App Store: на вашем iPhone или iPad запустите App Store.
- Поиск 1xBet: используйте строку поиска и введите «1xBet», чтобы найти приложение.
- Загрузите приложение 1xBet. Найдя приложение 1xBet, нажмите кнопку «Получить», чтобы начать загрузку приложения 1xBet в России. Приложение автоматически установится после завершения загрузки.
- Запустите приложение 1xBet: после установки вы увидите значок 1xBet на главном экране. Нажмите на него, чтобы открыть мобильное приложение 1xBet.
- Войдите или зарегистрируйтесь: Если у вас уже есть учетная запись в приложении 1xBet, вы можете войти в систему, используя свои учетные данные. Если нет, вы можете легко зарегистрироваться прямо через приложение 1x Bet.
Системные требования для iOS
Чтобы приложение 1xBet работало без проблем на устройствах iOS, рекомендуется соблюдать следующие системные требования:
- Версия iOS: 8.0+
- Оперативная память: 1 ГБ
- Процессор: 1,2 ГГц
- Внутреннее пространство: 90 Мб
Регистрация и подтверждение учетной записи в приложении 1xBet
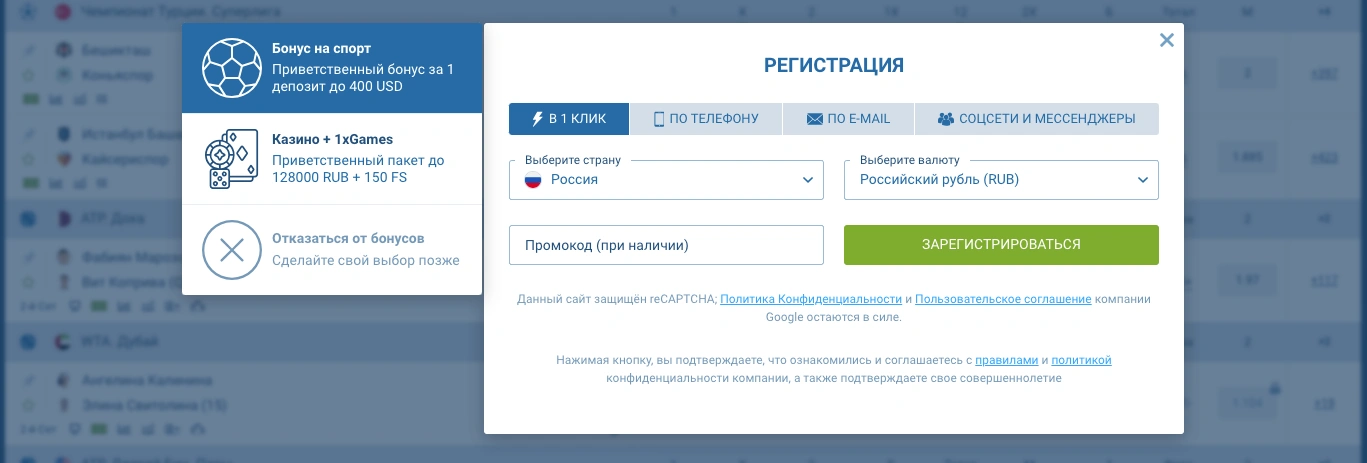
Регистрация аккаунта
- Загрузите и откройте приложение 1xBet. Установите приложение 1xBet на свое устройство Android или iOS и откройте его.
- Начать регистрацию: нажмите кнопку «Зарегистрироваться» на главном экране приложения.
- Введите данные: выберите способ регистрации (в один клик, телефон, электронная почта или социальная сеть) и укажите необходимую информацию.
- Установите пароль: создайте и подтвердите надежный пароль.
- Полная регистрация: отправьте форму после согласия с условиями.
Подтверждение аккаунта
- Подтвердите адрес электронной почты/телефон: проверьте свою электронную почту или SMS на наличие ссылки или кода подтверждения и следуйте инструкциям.
- Предоставить документы, удостоверяющие личность. При необходимости предоставьте документы, удостоверяющие личность, для проверки учетной записи.
- Подождите проверки: дайте 1xBet некоторое время для проверки вашей учетной записи.
- Активация учетной записи: вы получите уведомление, как только ваша учетная запись будет проверена и готова к использованию.
Обновление 1xBet Apk
- Посетите официальный сайт: регулярно проверяйте официальный сайт 1xBet на наличие обновлений.
- Скачать последний APK: Если доступно, загрузите новую версию APK с веб-сайта.
- Удалить предыдущую версию: удалите старую версию приложения со своего устройства.
- Установить новый APK: откройте загруженный файл и установите новую версию.
- Запустите приложение 1xBet: откройте обновленное приложение 1xBet и войдите в систему, чтобы подтвердить обновление.
Бонусы
Когда вы делаете свой первый депозит в приложении 1xBet, оно предлагает заманчивый стимул. Получите 100% бонус на свой первоначальный депозит с возможностью выиграть до 15 600 . Этот бонус эффективно удваивает вашу мощность ставок, позволяя вам изучить более широкий спектр вариантов ставок на спорт и увеличить ваши шансы на выигрыш. Приложение 1xBet предназначено не только для любителей спортивных ставок.

Игроки в казино могут насладиться солидным приветственным пакетом. Этот приветственный пакет включает в себя бонус до 100% на сумму до 150 000. Кроме того, он дополнен 150 бесплатными вращениями, которые идеально подходят для изучения разнообразия игровых автоматов, доступных в приложении. С помощью этого пакета вы можете окунуться во множество игр казино, от классических настольных игр до новейших игровых автоматов, улучшая свой игровой опыт за счет дополнительных средств и острых ощущений от бесплатных вращений.
Плюсы и минусы приложения 1xBet
| Плюсы приложения 1xBet | Минусы приложения 1xBet |
| Широкий выбор спортивных игр и игр казино. | Требует регулярных обновлений |
| Удобный и простой в навигации | Могут иметь региональные ограничения |
| Включает ставки в реальном времени и трансляции. | Может быть сложным для новичков |
| Многоязычный, включая несколько языков | |
| Разнообразие вариантов пополнения и вывода средств |
Приложение 1xBet в России предлагает комплексную и удобную платформу для любителей ставок на спорт и игр в казино. Благодаря широкому спектру вариантов ставок, функциям ставок в реальном времени и потоковой передачи, а также многоязычной поддержке, он хорошо удовлетворяет разнообразные потребности своих пользователей. Разнообразие способов оплаты еще больше повышает удобство для пользователей из России. Хотя необходимость регулярных обновлений и потенциальная сложность для новых пользователей являются незначительными недостатками, общая функциональность и преимущества приложения 1xBet делают его популярным выбором для онлайн-ставок в России. Его способность адаптироваться к местным предпочтениям, включая язык и варианты оплаты, подчеркивает его стремление предоставить персонализированный и доступный опыт ставок.
Популярные вопросы
На каких платформах доступно приложение 1xBet?
Приложение 1xBet доступно как для устройств на платформе Android, так и для устройств iOS (iPhone и iPad).
Какие особенности казино предоставляет 1xBet?
В разделе казино приложения 1xBet вы найдете разнообразные слоты, настольные игры (блэкджек, рулетка, баккара), а также игры в реальном времени с живыми дилерами.